Compressed Gas
Compressed gases are any materials or mixtures in containers having an absolute pressure exceeding 40 psi at 70°F (20°C) or exceeding 104 psi at 130°F (54.5°C). These cylinders are found in various locations across campus, especially at labs, medical buildings, and Facilities Services shops.
Handling of compressed gases may be considered more hazardous than the handling of liquid and solid materials because of the following properties: high pressure, ease of diffusion, low ignition points for flammable gases, low boiling points, and in some cases lack of visual and/or odor detection of hazardous gases. Because of these properties, failure to follow proper procedures can result in injury and property damage. Thus, employees handling compressed gases need to have the requisite knowledge to protect themselves against the hazards of this material.
Types of Compressed Gases
In general, there are three major types of compressed gases that are stored: liquefied, non-liquified and dissolved gases.
Liquefied gases – These are gases that can become liquids at normal temperature when they are inside cylinders under pressure. They exist inside the cylinder in a liquid-vapor balance or equilibrium. Examples are chlorine, carbon dioxide, anhydrous ammonia, liquefied nitrous oxide, liquefied petroleum gas (LPG), etc.
Non-Liquefied gases – Unlike liquefied gases, these gases do not become liquid when they are compressed at normal temperatures, even at very high temperatures. They are also known as compressed or pressurized gases. Common examples include nitrogen, oxygen, air, carbon dioxide, helium, argon, etc.
Dissolved gases – These are gases dissolved in a liquid phase solvent. Dissolved gas cylinders are packed with an inert, porous filter saturated with the solvent which stabilizes the volatile gas. The most common example is acetylene, which can explode at atmospheric pressure. These gases are very unstable. For example, acetylene cylinders have to be fully packed with an inert, porous filter that is saturated with acetone to prevent explosions. As acetylene is added to the cylinder, the gas dissolves in the acetone making the acetylene solution stable.
Types of Compressed Gas Cylinders
Compressed gas cylinders come in different shapes and designs, which are mostly based on the pressure of the gases they contain. In general, they are grouped as high-pressure cylinders, low-pressure cylinders, and cryogenic containers.
The high pressure cylinders are typically tall and narrow, thick-walled, heavy when empty, generally made of steel or aluminum, and can withstand up to 10,000 psi. Common examples are nitrogen, helium, hydrogen, oxygen, and carbon dioxide cylinders.
Low-pressure cylinders are typically fatter and lighter than high pressure; they have thin-walled, welded seams, and can withstand up to 500 psi. Common examples are liquefied petroleum gases (LPG, like propane) and refrigerant gases.
Cryogenic containers – these containers operate at a pressure of 20 – 500 psi. They have relief valves to help vent pressure as the temperature increases. Examples of cryogenic materials are nitrogen, oxygen, argon, carbon dioxide, etc.
Hazard of Compressed Gas
There are multiple hazards associated with compressed gases. They can be toxic (e.g. methyl bromide, anhydrous ammonia, chlorine, etc.), flammable (e.g. hydrogen, acetylene, etc.), corrosive (e.g. ammonia, chlorine, hydrogen chloride/ bromide, etc.) and oxidizing (e.g. nitrous oxide, oxygen, etc.). In contrast, some of them can be inert (chemically uncreative), non-flammable, and/or non-toxic. For example, nitrogen and carbon dioxide are non-flammable, while argon and helium are inert gases. Despite this property, they can still be harmful in some other ways. In the event of a leak, inert gases can quickly displace air in a large area creating an oxygen-deficient atmosphere, toxic gases can create poison atmospheres, and flammable or reactive gases can result in fire and explosion.
Besides the chemical characteristics of these gases, the cylinders on their own can be hazardous due to their physical size and weight. A gas cylinder can break containers and crush feet. If their valves are broken off, they can travel far and out of control causing significant injury and damage. Some of the specific hazards of compressed gases are listed below:
- Fire or explosion from the release of flammable gases near ignition sources (e.g. acetylene)
- Spontaneous combustion from oxidizing gases (e.g. nitrous oxide, oxygen)
- Toxic property for exposure to concentrations above permissible exposure limits (e.g anhydrous ammonia)
- Asphyxiation by displacement of oxygen (e.g. nitrogen)
- Leaks
- Incorrect storage
- Faulty equipment and/or connections
- Physical risks
- Manual handling
- Gas density
- Sudden release of gas if cylinder is damaged
- Pressure
Receiving
When cylinders are received in any location on campus, the responsible person should ensure the following -:
- Confirm cylinder content is the same as what was ordered.
- Visually inspect them for damage
- Confirm that cylinder(s) is/are properly labeled (see photo below).
- Cylinders delivered without labeling or identification should not be accepted.
- Do not rely on color as your primary method of identification. Cylinder color changes for different suppliers, which makes it unreliable as a method of identification
- If trained to do so, check that the valve is in place and can be easily removed. Each cylinder received from approved vendors is expected to come with a valve protection cap.
- Obtain or print the Safety Data Sheet and add it to your safety binder
Note – locations submitting chemical inventory to EH&S should include compressed gases information as well.

Image credit: Trainex
Storage
One of the most common questions we get asked at EH&S is how to properly store and secure compressed gas cylinders. We hope the information provided below will answer those questions. Please contact the EH&S office at (252) 328 -6166 or safety@ecu.edu if you have further questions.
Generally, Compressed gas cylinders should be stored in a level, cool, dry, well- ventilated, fire-resistant area that meets all applicable federal, state, and local regulations. Storage must also comply with Compressed Gas Association (CGA) standards.
General Storage Requirements
Store cylinders away from sources of ignition or excessive heat. They should be stored away from direct rays of the sun or any other excessive heat sources. Do not exposure cylinders to temperatures above 125oF. Pressure-relief devices are installed on most cylinders to prevent the rupture of a normally pressurized cylinder when it is inadvertently exposed to fire or high temperatures.
Store cylinders in an upright position, whether full or empty. Also, cylinders should always be secured in an upright position except, if necessary, for short periods of time while cylinders are actually being hoisted or carried.
Cylinders should not be stored near elevators or gangways, or in locations where heavy moving objects may strike or fall on them. They should be stored away from heavy traffic, emergency exits, or places that subject them to tampering by unauthorized persons. Do not store cylinders in hallways.
All cylinders must be secured to a wall, bench, or other fixed support, double-chained at 1/3 and 2/3 position of the cylinder height (see photo below). Cylinder stands (see photo below) are an alternative to straps. Note: storing on portable carts is not allowed, only for transport.
Cylinders must be individually strapped or secured to prevent falling or rolling.
Note: Bungee cords, zip ties, ropes, or wires are not acceptable means of securing cylinders.
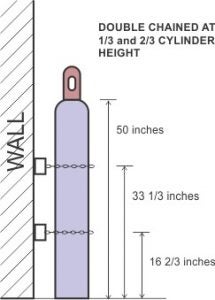

Image credit: hopkinssafety.edu Image credit: USAsafety.com
Other Storage Requirements
- Do not use a transport device or cart to hold a cylinder in use
- Names and hazard classes of the gases must be posted in the cylinder storage area.
- Segregate cylinders by hazard classes while in storage. When there is a need for mixed storage, store cylinders only with other compatible gases.
- Do not store flammable gases (e.g. acetylene) near oxidizers (e.g. oxygen) or combustible materials. Store oxygen (full or empty) away from flammable gases.
- Oxygen should be stored at least 20 feet away from flammable gases or separated by a 5ft. high non-combustible wall having a fire-resistance rating of at least one-half hour.
- Never store liquid or gas chlorine cylinders with ammonia cylinders.
- When cylinders are empty or not being used, ensure that the valve is closed, the regulator removed, and the valve protector cap is secured in place.
- Keep the number of cylinders in each location to a minimum to reduce physical (fire) and chemical (toxic gas exposure) hazards.
- Cylinders should not be placed where they might become part of an electrical circuit.
- Mark or tag empty cylinders “EMPTY”
- Full and empty cylinders should be stored separately in clearly marked areas. Objects should not be stored on top of gas cylinders
- For corrosive gases, gas distribution systems must be gas compatible. Corrosive gases should be stored for the shortest possible time to prevent corrosion of valves, labels, and regulators; and to also avoid potential leakage.
Use and Handling
Installation
Cylinders must be installed directly by supplier, Facilities Services trained staff, or lab personnel who are trained for such work.
All responsible users of compressed gas cylinders must be trained on proper use and handling techniques.
Untrained lab personnel should not install or tamper with compressed gas cylinders, including pressure relief valves and product labels.
Cylinder installation should be evaluated before use to ensure its operations would not introduce additional hazards to the area.
Before Use
Read all label information and the safety data sheet to be familiar with the property of the gas and the associated hazards.
Wear appropriate PPE – refer to the Safety Data Sheet
Leak test fittings using Snoop or soap solution. Never use an open flame to detect leaks.
Check piping and connections for integrity before use
Ensure cylinder valves are positioned in a way that is always accessible.
All compressed gas cylinders must be used with a pressure-regulating device to reduce the cylinder pressure to those recommended by the manufacturer.
During Use
Always use cylinders in well-ventilated areas.
Use appropriate regulator, compatible manifold materials, and pressure-rated tubing and piping for all applications. Do not alter, adapt, or use Teflon tape on regulators
All tubing should be periodically checked for integrity. Damaged tubing should be removed from service
Repair or alteration of compressed gas cylinders is prohibited
Cylinder should never be emptied to a pressure lower than 172kPa (25 psi/in2). Do not empty cylinder completely to prevent backflow of contaminants or air.
Valves, Regulators, and Piping
Valves
 The cylinder valve is a device used to contain the cylinder contents that is under pressure. They act as the primary safety mechanism
The cylinder valve is a device used to contain the cylinder contents that is under pressure. They act as the primary safety mechanism
Do not tamper with cylinder valves.
Valves should never be opened without a regulator attached.
Never open the cylinder valves unless the regulator is completely closed. Likewise, always turn off the cylinder by first closing the main cylinder valve and then the regulator. The pressure gauges should go back to zero
Always open the main cylinder valve slowly and avoid opening it all the way.
Do not face the gauge while opening a cylinder. Stand on the side in case of a malfunctioning valve. Cylinder valve must be closed when the cylinder is not in active use.
Do not use wrenches or pliers to open cylinder main valve. If the valve does not rotate, contact the supplier immediately
Regulator
 The regulator is another safety device that is fitted to a compressed gas cylinder. It reduces the cylinder’s high pressure to a safe operating working pressure. Proper regulator selection is therefore critical for both the safety and effectiveness of gas cylinders There are different types of regulators for different applications. They come as either single stage or two stages.
The regulator is another safety device that is fitted to a compressed gas cylinder. It reduces the cylinder’s high pressure to a safe operating working pressure. Proper regulator selection is therefore critical for both the safety and effectiveness of gas cylinders There are different types of regulators for different applications. They come as either single stage or two stages.
Regulators are designed to be fitted directly to the cylinder valves. No other fittings, connections, or lubricants should be used to connect a regulator to a gas cylinder valve
Never use a damaged or defective regulator. A regulator can explode, if faulty.
Do not operate a cylinder regulator if you have not been trained to do so. Always contact your supervisor or supplier if you have not been trained.
Piping
Do not use copper piping for acetylene
Do not use cast iron pipe for chlorine
Do not use oil or lubricants on equipment used with oxygen
Do not use Tygon and plastic piping for any portion of a high-pressure system
Keep connections to piping tight
Periodically inspect piping connections for leaks and damages
Ground all cylinders and piping containing flammable gases to static electricity hazards
When piping hydrogen, use stainless steel tubing to prevent the buildup and potential spark from static electricity.
All piping and equipment must meet the standards of the Compressed Gas Association
Transportation
All connected equipment (e.g. regulator, piping, etc.) must be removed before cylinder is transported
Ensure valve is in place and secure before handling cylinder. Valves must be closed
Wear appropriate PPE
Transport cylinder using a suitable cylinder cart or hand truck 
Move cylinders with safety cap in place
Do not lift or move cylinder by the cap
Do not drag, roll or slide cylinders
Do not drop or strike cylinders together
Always handle cylinders as if they were full Image credit: ccohs.ca
Do not handle cylinder alone if it is not safe to do so. Get help rather than suffer an injury
Transport one cylinder at a time. If a two cylinder cart is used, ensure cylinders are properly restrained before being transported
Only transport cylinder in a freight elevator, if available
Disposal
Close and tighten valves
Replace safety caps on cylinders
Store empty cylinders separately from full cylinders or those in-service. Note: A cylinder is empty at 25 psi
Contact supplier/vendor or your department’s contact person for instructions to return used cylinders
Note: Departments must include compressed gases in their annual inventory submitted to EH&S